Beaker (Nitrobenzene)
The printable version is no longer supported and may have rendering errors. Please update your browser bookmarks and please use the default browser print function instead.
Description
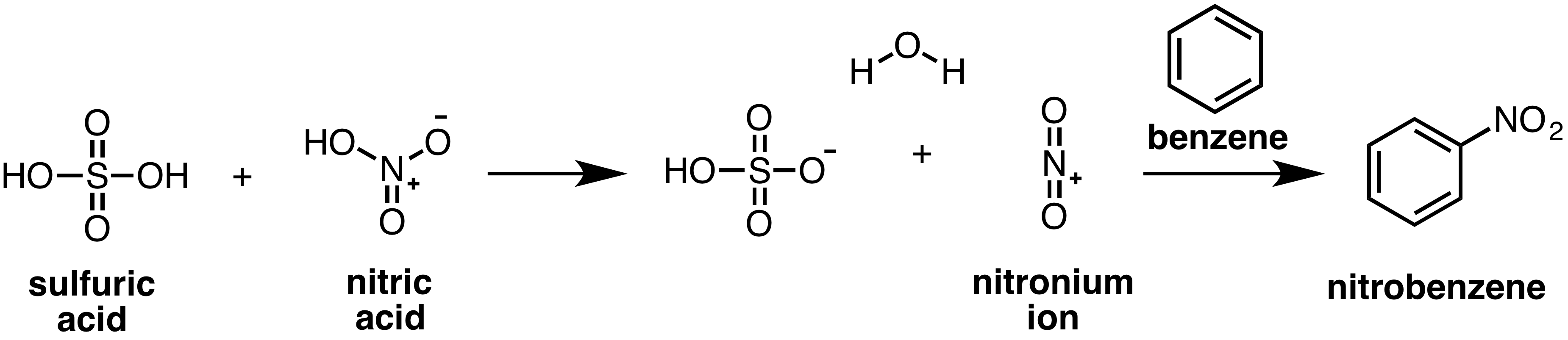
Nitrobenzene can be made through an electrophilic aromatic substitution reaction between benzene, nitric acid and sulfuric acid. Concentrated sulfuric acid and nitric acid react to generate a reactive nitronium (NO2+) ion which can then be attacked by a molecule of benzene.
External
Nitrobenzene on en.wikipedia.org
Properties
| Name | Value |
|---|---|
| Release Version | 1.1.2 |
Recipes
Crafting Table
| Outputs | Components | Recipe |
|---|---|---|
Chemical Processor
| Outputs | Components | Recipe |
|---|---|---|