Difference between revisions of "Vial (Nitrobenzene)"
Jump to navigation
Jump to search
(updated nitrobenzene reaction image size) |
|||
| (3 intermediate revisions by one other user not shown) | |||
| Line 1: | Line 1: | ||
==Description== | ==Description== | ||
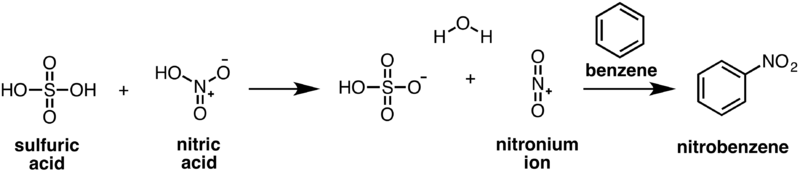
| − | Nitrobenzene can be made through an electrophilic aromatic substitution reaction between benzene, nitric acid and sulfuric acid. Concentrated sulfuric acid and nitric acid react to generate a | + | Nitrobenzene can be made through an electrophilic aromatic substitution reaction between benzene, nitric acid and sulfuric acid. Concentrated sulfuric acid and nitric acid react to generate a reactive nitronium (NO<sub>2</sub><sup>+</sup>) ion which can then be attacked by a molecule of benzene. |
| − | [[Image: | + | [[Image:Nitrobenzene-nitration.png|800px]] |
==External== | ==External== | ||
Latest revision as of 17:29, 2 November 2015
Description
Nitrobenzene can be made through an electrophilic aromatic substitution reaction between benzene, nitric acid and sulfuric acid. Concentrated sulfuric acid and nitric acid react to generate a reactive nitronium (NO2+) ion which can then be attacked by a molecule of benzene.
External
Nitrobenzene on en.wikipedia.org
Properties
| Name | Value |
|---|---|
| Release Version | 1.1.2 |
Recipes
Crafting Table
| Outputs | Components | Recipe |
|---|---|---|
Chemical Processor
| Outputs | Components | Recipe |
|---|---|---|